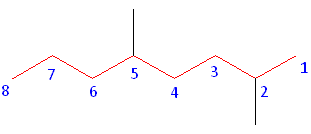
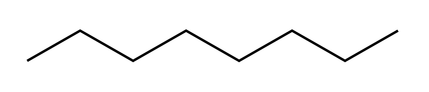
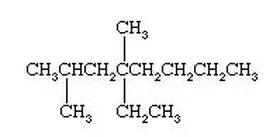
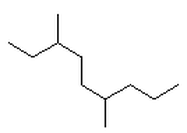
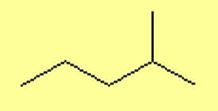
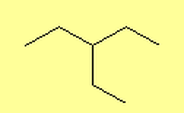
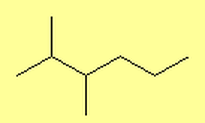
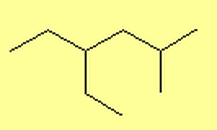
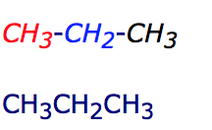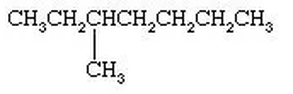What are Alkanes?
An alkane is a hydrocarbon in which all the carbon atoms are connected by single bonds. This gives them a general formula of CnH2n+2.
The general formula is used to calculate the number of hydrogens or carbons.
The general formula is used to calculate the number of hydrogens or carbons.
How to Name Alkanes:
- Find the longest chain of carbons. (It does NOT have to be straight.) This goes at the end of the name.
- Identify any side branches. These go just before the longest chain name.
- If a side chain occurs more than once, indicate how many time it occurs. This goes just before the branch name. di - two, tri = three, tetra = four. After that the prefixes are the same as for chain-length.
- Indicate which carbon the side-branch comes from by counting from the end of the main chain that gives the lowest possible number.
Prefixes:
•1carbon = meth
•2 Carbon = eth
•3 Carbon = prop
•4 Carbon = but
•5 carbon = pent
•6 Carbon = hex
•7 Carbon = Hept
•8 Carbon = oct
•9 Carbon = non
•10 carbon = dec
•1carbon = meth
•2 Carbon = eth
•3 Carbon = prop
•4 Carbon = but
•5 carbon = pent
•6 Carbon = hex
•7 Carbon = Hept
•8 Carbon = oct
•9 Carbon = non
•10 carbon = dec
How to Draw Diagrams:
Structural Diagrams:
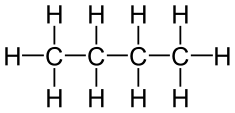
A structural diagram is the easiest to understand if you have never seen a chemical diagram before. It is also the most fundamental and basic diagram. To start off, you are going to place the carbon atoms in the middle, then afterwards you are going to place hydrogen atoms around it. This is because carbon atoms can bond with 4 other atoms while hydrogen can only bond with 2 other atoms.
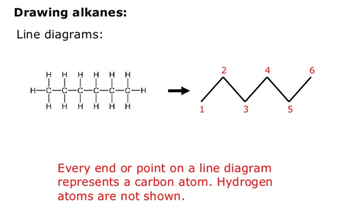
Line Diagrams (or skeletal):
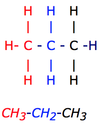
Condensed Diagrams:
|
1. As you can see, each section is color coded. The formula for the compound would be C4H8, but if you were to do a partially condensed formula, it would be CH3-CH2-CH3.
|
2. After you obtain the partially condensed formula which was CH3-CH2-CH3, you will have to remove the bonds from the formula and write it as CH3CH2CH3.
|
Molecular Diagrams:
C represents the number of carbon atoms and H represents the number of hydrogen atoms. This is different from condensed diagrams because you only use C and H together, and the number of the atoms are simplified.
Example 1
This is a very simple example with no branches. Since there are only 5 carbons and no carbons we call this pentane.
Example 2
Rule #1: Choose the longest, most substituted carbon chain containing a functional group. This example does not contain any functional groups, so we only need to be concerned with choosing the longest, most substituted carbon chain. The longest carbon chain has been highlighted in red and consists of eight carbons.
Rule #2: Carbons bonded to a functional group must have the lowest possible carbon number. If there are no functional groups, then any substitute present must have the lowest possible number. Because this example does not contain any functional groups, we only need to be concerned with the two substitutes present, that is, the two methyl groups. If we begin numbering the chain from the left, the methyls would be assigned the numbers 4 and 7. If we begin numbering the chain from the right, the methyls would be assigned the numbers 2 and 5. Therefore, to satisfy the second rule, numbering begins on the right side of the carbon chain as shown below. This gives the methyl groups the lowest possible numbering.
Rule 3: In this example, there is no need for the third rule. Because the two substitutes are identical, neither takes alphabetical priority with respect to numbering the carbons. This concept will become clearer in the following examples.
Example 3
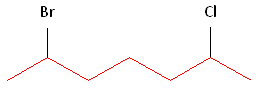
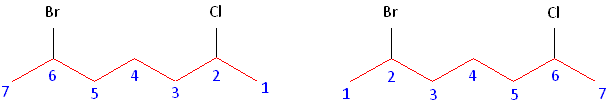
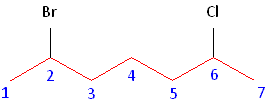
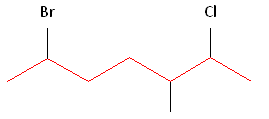
Rule #1: Choose the longest, most substituted carbon chain containing a functional group. This example contains two functional groups, bromine and chlorine. The longest carbon chain has been highlighted in red and consists of seven carbons.
Rule #2: Carbons bonded to a functional group must have the lowest possible carbon number. If there are no functional groups, then any substitute present must have the lowest possible number. In this example, numbering the chain from the left or the right would satisfy this rule. If we number the chain from the left, bromine and chlorine would be assigned the second and sixth carbon positions, respectively. If we number the chain from the right, chlorine would be assigned the second position and bromine would be assigned the sixth position. In other words, whether we choose to number from the left or right, the functional groups occupy the second and sixth positions in the chain. To select the correct numbering scheme, we need to utilize the third rule
Rule #3: After applying the first two rules, take the alphabetical order into consideration. Alphabetically, bromine comes before chlorine. Therefore, bromine is assigned the second carbon position, and chlorine is assigned the sixth carbon position.
Example 4
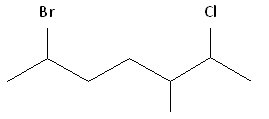
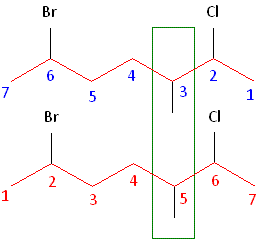
Rule #1: Choose the longest, most substituted carbon chain containing a functional group. This example contains two functional groups, bromine and chlorine, and one substitute, the methyl group. The longest carbon chain has been highlighted in red and consists of seven carbons.
Rule #2: Carbons bonded to a functional group must have the lowest possible carbon number. After taking functional groups into consideration, any substitutes present must have the lowest possible carbon number. This particular example illustrates the point of difference principle. If we number the chain from the left, bromine, the methyl group and chlorine would occupy the second, fifth and sixth positions, respectively. This concept is illustrated in the second drawing below. If we number the chain from the right, chlorine, the methyl group and bromine would occupy the second, third and sixth positions, respectively, which is illustrated in the first drawing below. The position of the methyl, therefore, becomes a point of difference. In the first drawing, the methyl occupies the third position. In the second drawing, the methyl occupies the fifth position. To satisfy the second rule, we want to choose the numbering scheme that provides the lowest possible numbering of this substitute. Therefore, the first of the two carbon chains shown below is correct.
Therefore, the first numbering scheme is the appropriate one to use.
Once you have determined the correct numbering of the carbons, it is often useful to make a list, including the functional groups, substitutes, and the name of the parent chain.
Rule #3: After applying the first two rules, take the alphabetical order into consideration. Alphabetically, bromine comes before chlorine. Therefore, bromine is assigned the second carbon position, and chlorine is assigned the sixth carbon position.
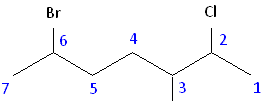
Parent chain: heptane 2-Chloro 3-Methyl 6-Bromo
6-bromo-2-chloro-3-methylheptane
Rule #3: After applying the first two rules, take the alphabetical order into consideration. Alphabetically, bromine comes before chlorine. Therefore, bromine is assigned the second carbon position, and chlorine is assigned the sixth carbon position.
Parent chain: heptane 2-Chloro 3-Methyl 6-Bromo
6-bromo-2-chloro-3-methylheptane
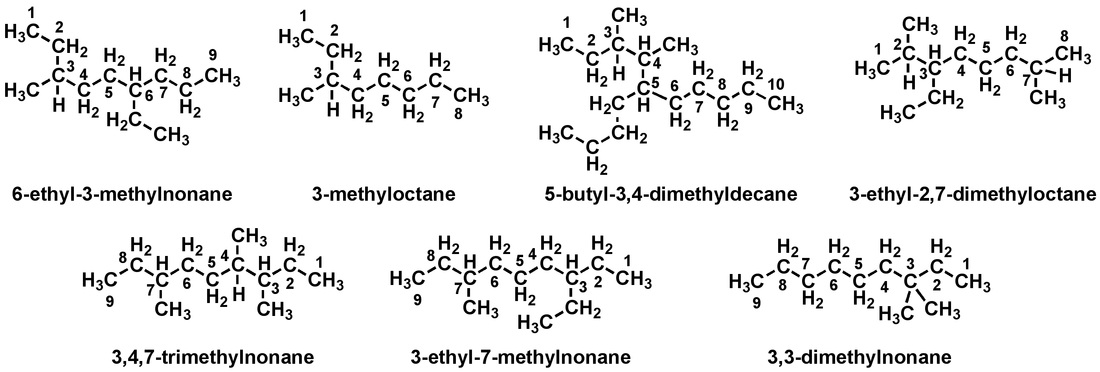
More examples!
Test Your Understanding
Draw the following formulas using a line formula.
- 2-methylpentane
- 3-ethylpentane
- 2,3-dimethylhexane
- 4-ethyl-2-methylhexane
Answers, don't cheat!
Answers for the drawings:
1. Octane
2. 3-methylheptane
3. 4-ethyl-2,4-dimethyloctane
4. 3,6-dimethylonane
1. Octane
2. 3-methylheptane
3. 4-ethyl-2,4-dimethyloctane
4. 3,6-dimethylonane
What are their everyday uses?
•Methane- natural gas
•Ethane – natural gas
•Propane – fuel for your BBQ
•Butane – lighter gas
•Pentane - blowing agents used in the production of polystyrene foam
•Hexane - formulation of glues for shoes
•Heptane – non-polar solvent used for getting rid of grease spots
•Octane – byproducts of gasoline (octane rating)
•Ethane – natural gas
•Propane – fuel for your BBQ
•Butane – lighter gas
•Pentane - blowing agents used in the production of polystyrene foam
•Hexane - formulation of glues for shoes
•Heptane – non-polar solvent used for getting rid of grease spots
•Octane – byproducts of gasoline (octane rating)