What are Alkenes?
Alkenes are a family of hydrocarbons containing a carbon-carbon double bond.
General formula is: CnH2n
Unsaturated: Has one or more double bonds in the chemical formula.
Saturated: Has only single bonds
General formula is: CnH2n
Unsaturated: Has one or more double bonds in the chemical formula.
Saturated: Has only single bonds
Examples
Examples of Cycloalkenes:
How to Name Alkenes:
1. Name the parent hydrocarbon by locating the longest carbon chain that contains the double bond and name it according to the number of carbons with the suffix -ene.
2. Number the carbons of the parent chain so the double bond carbons have the lowest possible numbers.
3. Write out the full name, numbering the substituents according to their position in the chain (alphabetical order)
4. . Indicate the double bond by the number of the first alkene carbon
5. If more than one double bond is present, indicate their position by using the number of the first carbon of each double bond and use the suffix -diene
2 double bonds: -triene
3 double bonds: -tetraene
2. Number the carbons of the parent chain so the double bond carbons have the lowest possible numbers.
3. Write out the full name, numbering the substituents according to their position in the chain (alphabetical order)
4. . Indicate the double bond by the number of the first alkene carbon
5. If more than one double bond is present, indicate their position by using the number of the first carbon of each double bond and use the suffix -diene
2 double bonds: -triene
3 double bonds: -tetraene
Practice
Name these structural diagrams:
Answers:
a) Ethene
b) Propene
c) Hexene
Draw the structural formulas for these alkenes:
a)Octene
b) Butene
c) Methene
Answers:
Octene
Butene
Trick question: Methene does not exist because an alkene requires a carbon-carbon double bond.
Types of Alkenes in Everyday Life:
-Ethylene is found in polyethylene, which is the basis of plastic bags and boxes
-Orlon is the result of Acrylonitril (propenenitril) and are found in clothes and synthetic textile materials
What are Alkynes?
Alkynes are hydrocarbons that contain carbon-carbon double bonds. The general formula is CnH2n-2.
Alkynes are unsaturated because they contain at least one carbon-carbon double bond, which is the keypoint between the difference of saturated and unsaturated.
Unsaturated: Has one or more double bonds in the chemical formula.
Saturated: Has only single bonds in the chemical formula.
Alkynes are unsaturated because they contain at least one carbon-carbon double bond, which is the keypoint between the difference of saturated and unsaturated.
Unsaturated: Has one or more double bonds in the chemical formula.
Saturated: Has only single bonds in the chemical formula.

Propyne
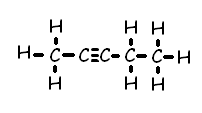
2-Pentyne
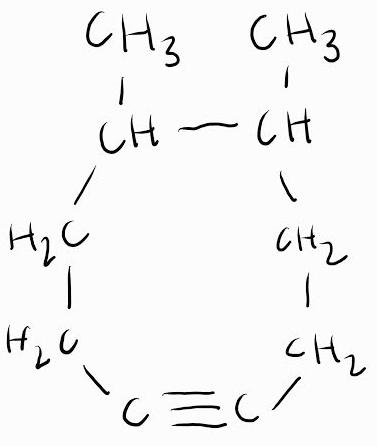
5,6-dimethyl-1-cyclooctyne
Practice Questions:
Name these Alkynes:
1. CH3CH2CH2C≡CCH2CH2CH2CH3
2. CH≡C-CH2-CH2-CH2-CH2-CH2-CH3
3. CH3-C≡C-CH2-CH2-CH2-CH3
Draw these Alkynes:
4. Ethyne
5. Butyne
6. Propyne
Name these Alkynes:
1. CH3CH2CH2C≡CCH2CH2CH2CH3
2. CH≡C-CH2-CH2-CH2-CH2-CH2-CH3
3. CH3-C≡C-CH2-CH2-CH2-CH3
Draw these Alkynes:
4. Ethyne
5. Butyne
6. Propyne
Answers (Don't Cheat!):
1. 4-nonyne
2. 1-octyne
3. 2-heptyne
1. 4-nonyne
2. 1-octyne
3. 2-heptyne
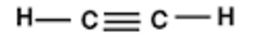
4.
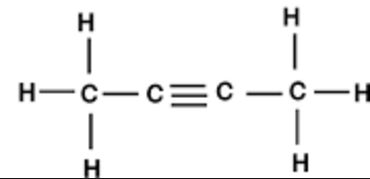
5.
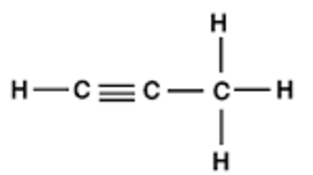
6.







